来源:JAMA. 2023 Jul 17. doi: 10.1001/jama.2023.13239. 1209
John R Sims, Jennifer A Zimmer, Cynthia D Evans, Ming Lu, Paul Ardayfio, JonDavid Sparks, Alette M Wessels, Sergey Shcherbinin, Hong Wang, Emel Serap Monkul Nery, Emily C Collins, Paul Solomon, Stephen Salloway, Liana G Apostolova, Oskar Hansson, Craig Ritchie, Dawn A Brooks, Mark Mintun, Daniel M Skovronsky; TRAILBLAZER-ALZ 2 Investigators
Importance: There are limited efficacious treatments for Alzheimer disease.
Objective: To assess efficacy and adverse events of donanemab, an antibody designed to clear brain amyloid plaque.
Design, setting, and participants: Multicenter (277 medical research centers/hospitals in 8 countries), randomized, double-blind, placebo-controlled, 18-month phase 3 trial that enrolled 1736 participants with early symptomatic Alzheimer disease (mild cognitive impairment/mild dementia) with amyloid and low/medium or high tau pathology based on positron emission tomography imaging from June 2020 to November 2021 (last patient visit for primary outcome in April 2023).
Interventions: Participants were randomized in a 1:1 ratio to receive donanemab (n = 860) or placebo (n = 876) intravenously every 4 weeks for 72 weeks. Participants in the donanemab group were switched to receive placebo in a blinded manner if dose completion criteria were met.
Main outcomes and measures: The primary outcome was change in integrated Alzheimer Disease Rating Scale (iADRS) score from baseline to 76 weeks (range, 0-144; lower scores indicate greater impairment). There were 24 gated outcomes (primary, secondary, and exploratory), including the secondary outcome of change in the sum of boxes of the Clinical Dementia Rating Scale (CDR-SB) score (range, 0-18; higher scores indicate greater impairment). Statistical testing allocated α of .04 to testing low/medium tau population outcomes, with the remainder (.01) for combined population outcomes.
Results: Among 1736 randomized participants (mean age, 73.0 years; 996 [57.4%] women; 1182 [68.1%] with low/medium tau pathology and 552 [31.8%] with high tau pathology), 1320 (76%) completed the trial. Of the 24 gated outcomes, 23 were statistically significant. The least-squares mean (LSM) change in iADRS score at 76 weeks was -6.02 (95% CI, -7.01 to -5.03) in the donanemab group and -9.27 (95% CI, -10.23 to -8.31) in the placebo group (difference, 3.25 [95% CI, 1.88-4.62]; P < .001) in the low/medium tau population and -10.2 (95% CI, -11.22 to -9.16) with donanemab and -13.1 (95% CI, -14.10 to -12.13) with placebo (difference, 2.92 [95% CI, 1.51-4.33]; P < .001) in the combined population. LSM change in CDR-SB score at 76 weeks was 1.20 (95% CI, 1.00-1.41) with donanemab and 1.88 (95% CI, 1.68-2.08) with placebo (difference, -0.67 [95% CI, -0.95 to -0.40]; P < .001) in the low/medium tau population and 1.72 (95% CI, 1.53-1.91) with donanemab and 2.42 (95% CI, 2.24-2.60) with placebo (difference, -0.7 [95% CI, -0.95 to -0.45]; P < .001) in the combined population. Amyloid-related imaging abnormalities of edema or effusion occurred in 205 participants (24.0%; 52 symptomatic) in the donanemab group and 18 (2.1%; 0 symptomatic during study) in the placebo group and infusion-related reactions occurred in 74 participants (8.7%) with donanemab and 4 (0.5%) with placebo. Three deaths in the donanemab group and 1 in the placebo group were considered treatment related.
Conclusions and relevance: Among participants with early symptomatic Alzheimer disease and amyloid and tau pathology, donanemab significantly slowed clinical progression at 76 weeks in those with low/medium tau and in the combined low/medium and high tau pathology population.
Trial registration: ClinicalTrials.gov Identifier: NCT04437511.
免责声明:相关信息仅限药物研发参考使用,本网站不保证信息真实和准确!
关注“药研苑”公众号,查看前景解析。

|
推荐阅读: ・2025年1-3季度口崩片市场哪个强? ・2025年1-3季度口溶膜制剂哪家强? ・免疫检查点抑制剂,谁将成为下一个王者? ・质子泵相关抑酸类药物市场即将回暖 ・中药1类新药距离封神还有多远 |
|
“药品营销避坑”试读: ・药品立项需要注意什么(一) ・药品生命周期管理(一) ・改剂型,口服液体制剂“金矿”还是“陷阱 |
我们提供如下咨询服务:药品信息发布、药品立项、市场前景分析、医院及药品零售市场分析、药品市场调研及制定推广策略、国外药品引进、国内批文转让、上市前后临床试验设计、药品彩页及主图设计、药品推广PPT制作。您可以关注公众号“药研苑”后,在主页面发送消息,咨询相关服务。


2024年10月29日,礼来Eli Lilly宣布Donanemab(商品名:Kisunla)TRAILBLAZER-ALZ 6研究3b期临床试验取得积极结果。
发布日期:2024-10-29 浏览数:1567
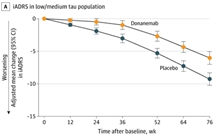
2023年7月17日Eli Lilly公布了抗β淀粉样蛋白(Aβ)单克隆抗体Donanemab的3期临床试验TRAILBLAZER-ALZ 2试验结果。试验显示 Donanemab能够有效改善Aβ斑块检测阳性阿尔茨海默症早期症状患者的认…
发布日期:2023-07-17 浏览数:1509

2024年7月2日,礼来公司宣布FDA批准Donanemab(商品名Kisunla™)350 mg/20 mL,每月一次静脉输注用于治疗处于轻度痴呆阶段,伴有轻度认知障碍(MCI),并经淀粉样蛋白病理学确诊的成人早期症状性…
发布日期:2024-07-02 浏览数:1310
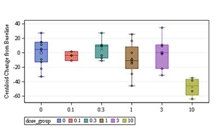
Intravenous donanemab 10 mg/kg can reduce amyloid deposits in AD despite having a shorter than expected half-life.
发布日期:2021-02-14 浏览数:1224
 改变滴定方案礼来降低其阿尔茨海默症抗体药物脑水肿发生率
改变滴定方案礼来降低其阿尔茨海默症抗体药物脑水肿发生率横切线®为注册商标
Copyright 2020 横切线®药研苑 备案号:粤ICP备18041379号-3